Navigation auf uzh.ch
Navigation auf uzh.ch
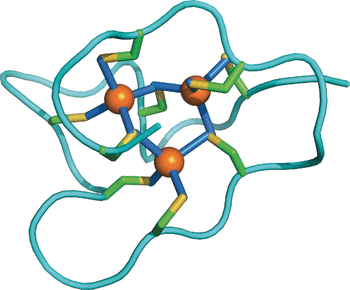
Snails are known for their intriguing ability to accumulate and detoxify heavy metals. They detoxify cadmium by binding it to metallothioneins, a class of small proteins rich in the sulfur-containing cysteine amino acid. These metallothioneins have an extraordinarily large capability for binding heavy metals. Oliver Zerbe and his co-authors have now discovered that the marine common periwinkle contains the largest version of the protein found yet, with one additional cadmium-binding domain and a one-third higher detoxification capacity. Studying the molecular differences between the proteins among various species, the scientists solved the solution structure of the periwinkle metallothionein by using nuclear magnetic resonance techniques and compared it with other known structures and sequences. Surprisingly, the periwinkle’s protein comprises three independent domains, while other known metallothioneins have only one or two. This feature may help the snail to survive in heavy-metal-polluted environments.
Link to the article and the press release of "Angewandte Chemie"