Organic Photoreleasing Systems
Our group develops organic photoreleasing systems actuated by near-infrared (NIR) or short-wave infrared (SWIR) light to facilitate delivery and release of multiple therapeutic agents simultaneously under biological settings. Such approach will enable the discovery and harnessing synergistic therapeutic effects, or triggering and studying fundamental biological processes by light. The research of our group lies at the interface of organic and physical chemistry, photochemistry and biochemistry. Our aim is to re-engineer small organic, transition-metal-free dyes into systems which can undergo direct cleavage of bonds with a single photon in the NIR or SWIR region. In parallel, we further employ the understanding of these systems to advance the area of bioimaging and biosensing, as diagnostics and therapy go hand-in-hand in eradicating the diseases which plague the modern society.
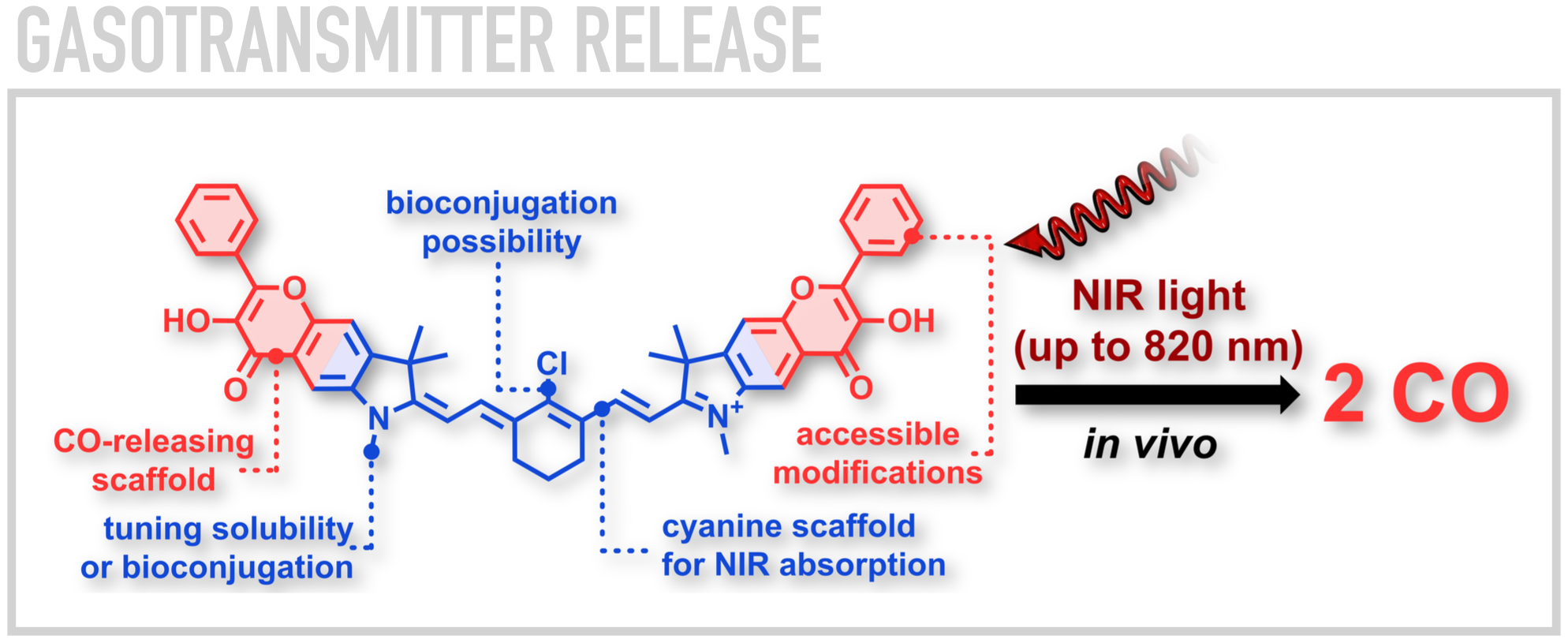
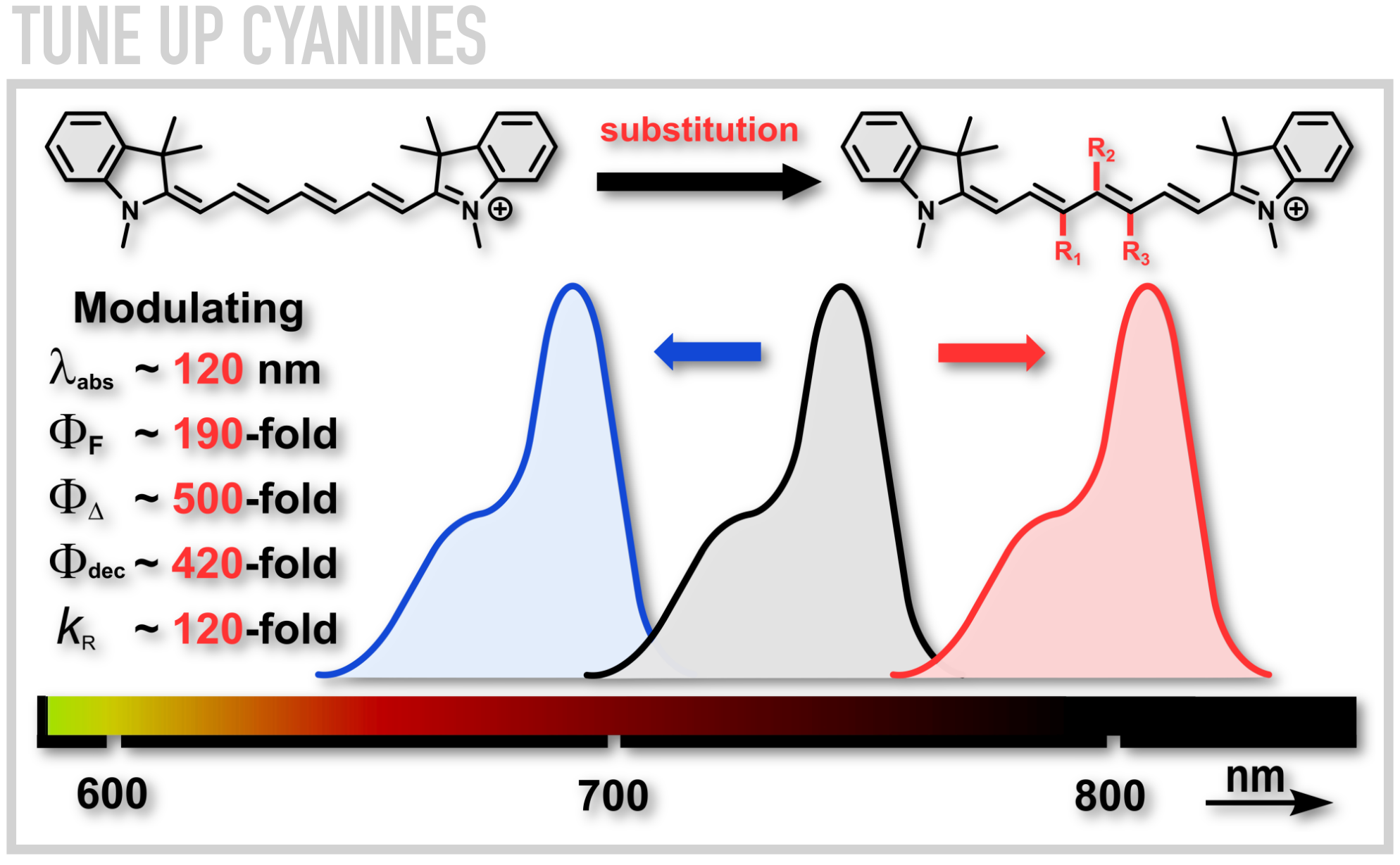
One type of molecules we investigate are cyanines, the relatives of FDA-approved indocyanine green dye prevalent in the contemporary medicine. Recently, we reported a methodology for installation of various substituents and patterns on the central polymethine backbone utilizing Zincke chemistry. The ability to modulate the photophysical and photochemical properties of cyanines within 2-3 orders of magnitude by a precise choice of substituent and its position within the chain has emerged as a consequence. We also designed cyanines which can deliver and efficiently release carbon monoxide, a naturally occurring signaling molecule, in mice upon exposure to NIR light at 820 nm at doses comparable to those employed in photodynamic therapy.