Renewable Energy and Sustainable Chemistry
New materials for photoelectrochemical (PEC) water splitting
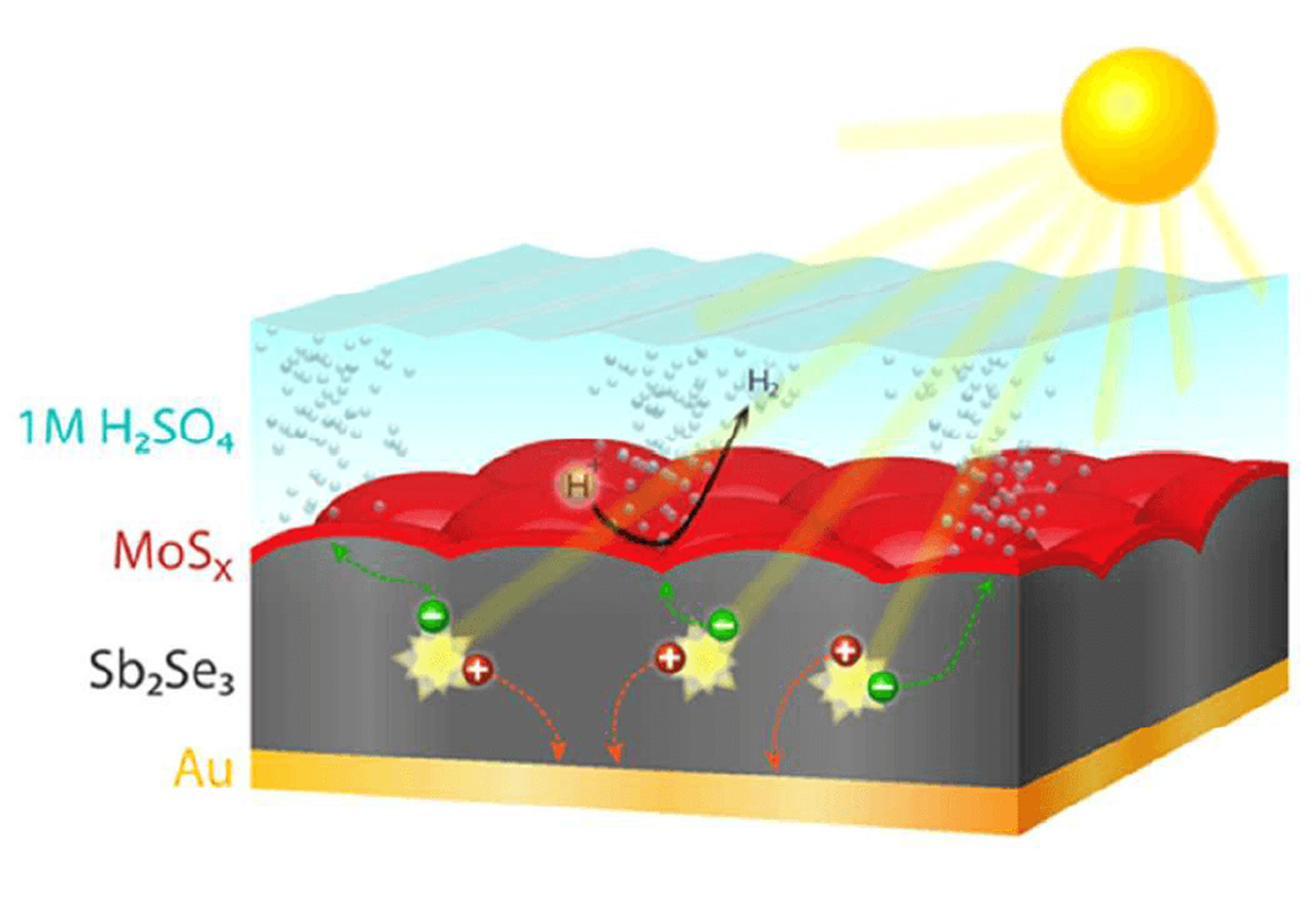
Particle-based PEC water splitting has the potential to produce solar hydrogen at a lower cost than fossil fuel-derived hydrogen, and is therefore a disruptive technology that could free us from a fossil fuel-based economy. However, new materials are needed to achieve the stability and efficiencies that are required for low cost hydrogen production. The first research theme of our group is the development of new thin film semiconductor materials, which could be adapted to particulate systems in future research. One promising material of our interest is Sb2Se3, a new material that we found is intrinsically stable in acidic solutions under full solar illumination. It is a strong candidate for a practical, scalable photocathode material if the efficiency can be improved.
Advanced analytical techniques for PEC water splitting
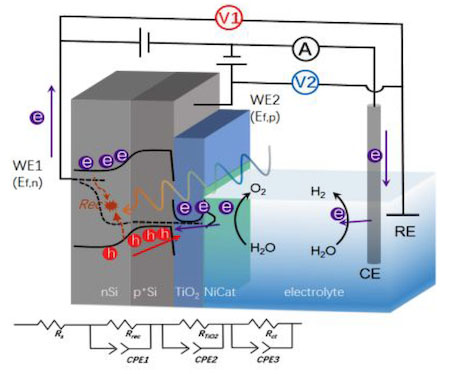
Studying semiconductor materials as thin films enables the use of a suite of techniques for in-depth characterization and understanding, leading to the identification of limitations and subsequent improvement of the system. The second line of our research involves the use of advanced analytical techniques to gain a complete understanding of the underlying (photo)physical processes in PEC materials, which is quite complex. We wish to further develop our novel implementation of the dual working electrode technique and the electrochemical impedance-based technique, as well as transient photocurrent and photovoltage studies using our newly built setup. Our aim is to find a general (photo)physical model that can be applied across a wide range of PEC systems, for fast identification of the limitations and rapid improvement in these systems.
Organic electrosynthesis
The main aim of this research is to advance the use of electrochemistry in organic synthesis, with the long-term goal of electrifying the chemical industry. The use of electrochemistry can prevent the use of dangerous, toxic, or expensive chemicals and materials, resulting in greatly reduced (toxic) waste production. Moreover, renewable sources of electricity can be used, which could eliminate the reliance on fossil fuels for energy in the chemical industry. The challenge is to merge the fields of organic chemistry and electrochemistry, which have traditionally evolved along separate lines.