Research
Research Interests
General
The research interests of the group include: high resolution laser spectroscopy; molecular dynamics; surface science, in particular in the production and growth mechanisms of electronic thin films; and numerical quantum chemistry. The central drive of the research is the structure and dynamics of molecules, in particular, nonradiative and chemical-decay mechanisms on a molecular level and the application of this to atmospheric chemistry.
Special topics
Photofragment Translational Spectroscopy
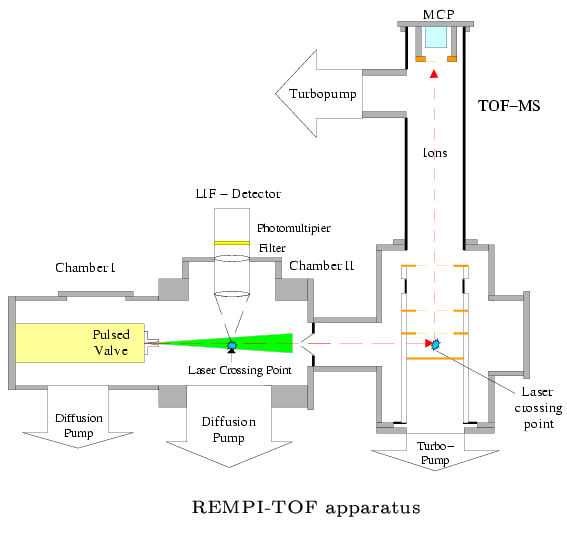
Photodissociation with LIF/REMPI-TOF Fragment Probing
While investigation of photodissociation processes using the time-of-flight method is very versatile, rotational resolution or beyond, and hence determination of rotational fragment state distributions, can usually not be achieved. Although much less general, since one is confined to well-known fragment species, fragment probing by LIF and REMPI-TOF yields very high energy resolution and hence rotational, vibrational and translational energy distributions, including special information of the dissociation process. Especially the measurements of partial cross sections provides an unprecedented insight into the microscopic decay of a molecule, and also serves as a test for quantum mechanical ab initio PES calculations and wave packet dynamics.
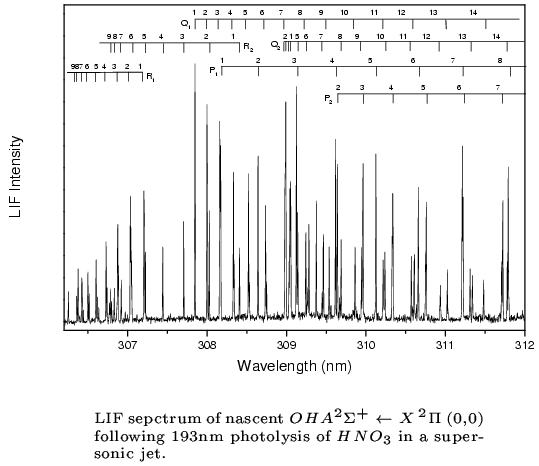
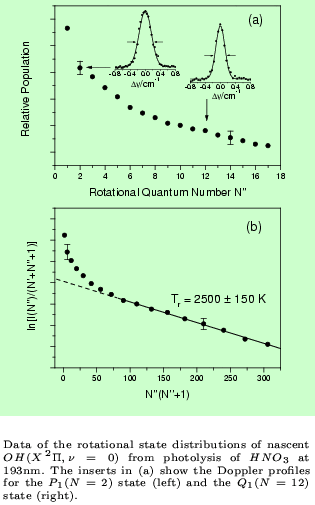
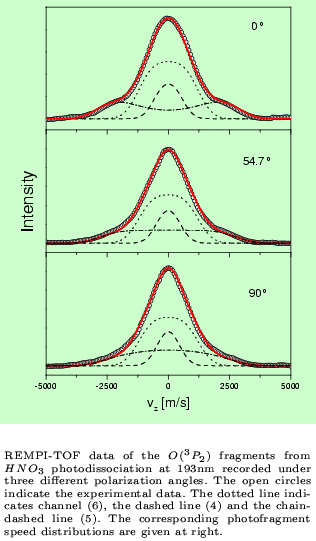
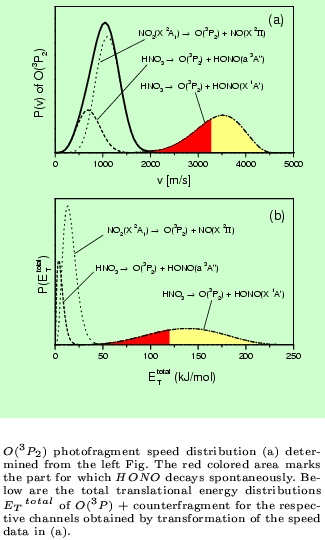
An example for a REMPI-TOF and LIF investigation is the photodissociation of
HNO3 at 193 nm. (Chem.Phys.Lett. 334 (2001) 39)
At this excitation wavelengths the following decay channels were found to be
active:
The combination of these two powerful detection methods provides e.g. the results shown in the figures below.
The photodissociation of water-clustered HNO3 studied at 193 nm by the LIF method
We studied the photodissociation of water-clustered HNO3 molecules at 193 nm, a process relevant in atmospheric chemistry, by generating water clusters by adiabatic expansion and doping them with HNO3 using the pickup method. The nascent OH(X2π) photofragments were probed by LIF, and the rotational state distribution in the exclusively produced vibrational ground state determined. These experiments were complemented by corresponding ones with argon and methanol clusters. The average cluster size was estimated to be ~ η 200 - 800 depending on the compound. The results of this study are compared with those previously obtained from the photodissociation of the HNO3 monomer.
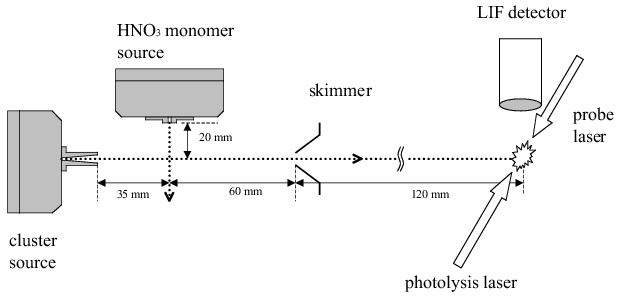
Fig.1: Experimental setup for cluster production by the pickup method and cluster photodissociation with LIF fragment detection.
Quantum beat spectroscopy is based on the excitation of a coherent superposition of eigenstates and the observation of their temporal evolution. In the most simple situation of two coherently excited eigenstates the fluorescence decay exhibits a superimposed oscillation, or quantum beat, whose frequency directly yields the energy separation between the eigenstates. The application of quantum beat spectroscopy is most rewarding when splittings smaller than the Doppler width are measured. Accordingly, splittings of rovibronic states which are induced by weak external fields, electric or magnetic, or by internal fields, such as in fine and hyperfine interaction, are conveniently assessed and structural parameters such as electric dipole moments, rotational constants, Landé g-factors, and hyperfine constants have been determined. Furthermore, quantum beats are a powerful tool for studying intramolecular relaxation in polyatomic molecules and its underlying mechanism.

Molecular beam apparatus for quantum beat measurements
Measurement of the Majorana effect in a molecule using a switched magnetic field and quantum beat detection
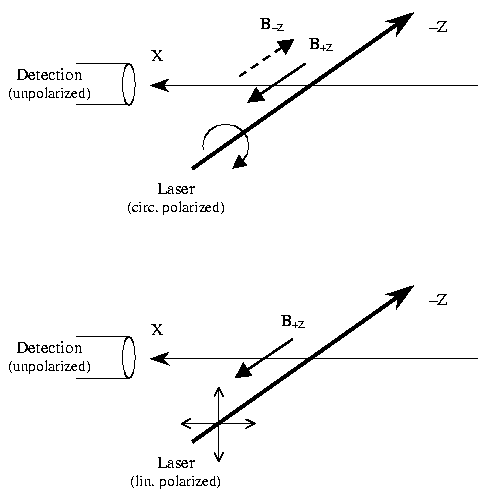
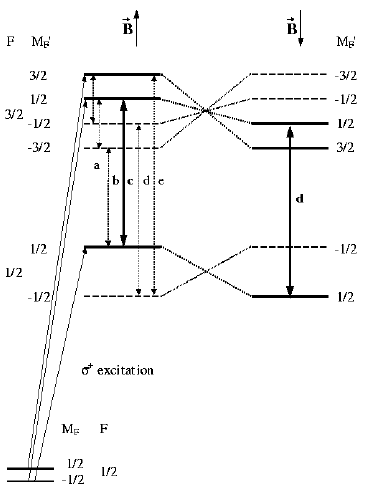
By means of quantum beat spectroscopy we investigated the dynamics
of magnetic moments associated with hyperfine levels in the molecule
13CS2 under "spin-flip conditions". Oriented
molecules in a cold beam were prepared by excitation with a circularly
polarized laser pulse (~ 3 ns) in a weak magnetic field (B = 5.4 G).
Subsequently they were exposed to a rapid field inversion which left
the magnetic moment in its initial orientation. The initially created
coherences among the excited hf levels were conserved after field
reversal and thus were explored to characterize the changes in the level
structure due to this "Majorana spin-flip" process.
Hubertus Ring et al.
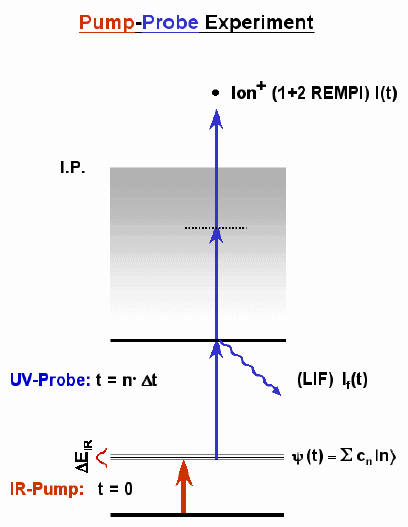
Time-resolved Double Resonance Spectroscopy
Investigation of the dynamics of molecules in the electronic ground state is of utmost importance for the understanding of chemical reactions. Information about the ground state dynamics is obtained from frequency domain spectra (e.g. absorption), but due to line broadening and congestion, it is difficult to interpret. We therefore follow the evolution of the states excited with a pulsed IR laser by monitoring the laser-induced fluorescence after time-delayed pulsed UV excitation (time-resolved IR-UV double resonance). Monitoring the time evolution of coherently excited S0 states, quantum beat spectroscopy is applicable to the ground state. The advantages of this technique are lifetime-limited resolution of coherently excited states (see quantum beat spectroscopy) and simplification of frequency domain spectra. A variety of experiments, including step-wise excitation, V-type double resonance schemes, and fluorescence dip spectroscopy can be carried out with our present setup as well.
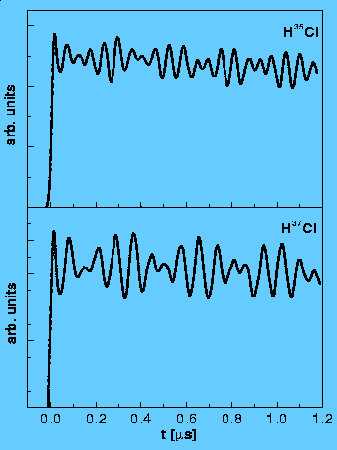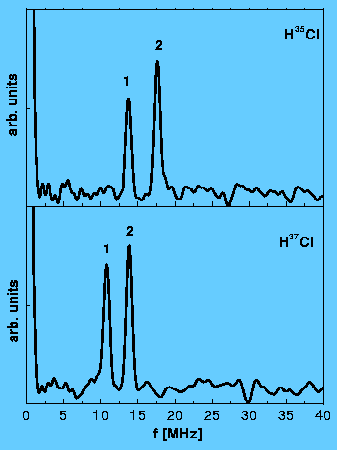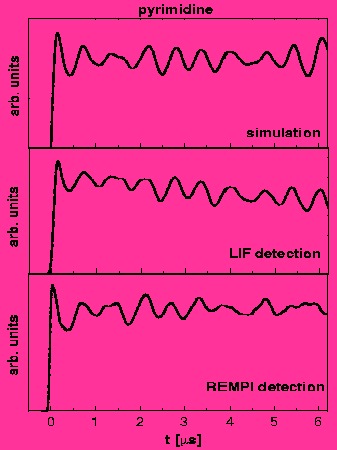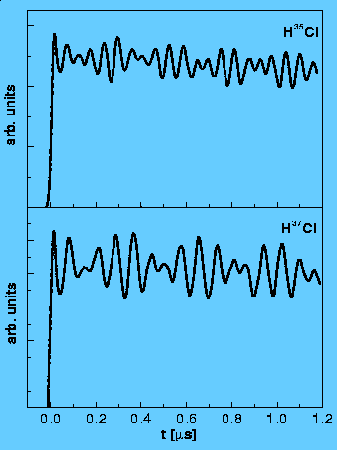
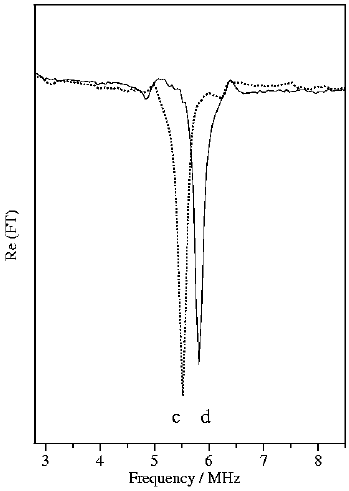
Nuclear quadrupole hyperfine quantum beats in HCl and pyrimidine measured by the pump-probe method using REMPI/LIF detection. H. Lammer, R.T. Carter and J. R. Huber
Surface Science Laboratory
In the surface science laboratories, fundamental aspects of film growth and gas-phase and heterogeneous surface chemistry are investigated. A novel laser ablation technique incorporating a synchronized pulsed gas source has been developed for the growth of oxide and nitride films [1,2]. This technique has been shown to have significant advantages over conventional laser ablation methods [3]. Our approach to a deeper understanding of the microscopic processes at play is shown schematically in Fig. 1. The ablation and "blow-off" characteristics of a variety of metals under irradiation from 248 nm light have been characterized [4]. The interaction between the resulting ablation plume of the metallic species with a supersonic expansion were then further investigated for the system of a Ti plasma and a nitrogen gas pulse [3]. The mass spectra of the resulting species for different delays between the gas pulse and laser pulse trigger are shown in Fig. 2. It was found that if the conditions were carefully chosen, epitaxial TiN(001)[100] || Si(001)[100] films could be grown at temperatures as low as 200o C. The growth of the films could be monitored in-situ using reflection high-energy electron-diffraction (RHEED), as shown in Fig. 3. Growth rates could be monitored by RHEED oscillations, while the in-plane growth mechanisms during interface formation could be determined by close examination of the RHEED patterns, given in Fig. 4 for growth at 730o C [5]. Further work will include aspects of film growth of other simple oxides and nitrides such as c-BN and GaN, plus the use of RHEED to characterize non-perfect catalytic surfaces in order to gain a fuller understanding of the processes involved in heterogeneous catalysis.
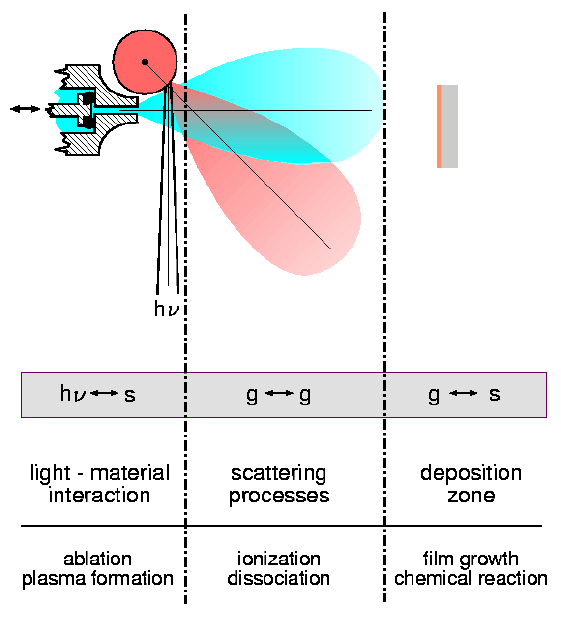
For details see: J. of Materials Chemistry 12 (2002) 397 , Rev. Mod. Phys. 72(2000)34






